The FDA has announced another nationwide recall of a high blood pressure medication, due to a potentially “life-threatening” labeling mixup. Healthcare is voluntarily recalling a single lot of 12.5-milligram hydrochlorothiazide tablets, after a pharmacy reported finding a 100-count bottle that contained spironolactone tablets, 25mg. Spironolactone tablets are prescribed to treat cirrhosis of the liver, treat congestive heart failure, and other medical conditions. According to the FDA, the rest of the lot (PW05264) has been recalled for the potential mix -up of labeling, but Accord Healthcare stated the remainder of its drugs are unaffected.
Videos by Rare
Both spironolactone and hydrochlorothiazide can be used to treat high blood pressure. Yet, spironolactone can cause the body to flush out excess water and sodium and store potassium, which is why the Food and Drug Administration warns patients who mistakenly take it instead of hydrochlorothiazide after they could experience hyperkalemia or elevates potassium levels. The effects of hyperkalemia range from “limited health consequences to a severe life-threatening situation in certain individuals.”
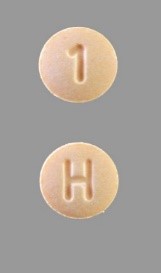
As of now, there have been no reports adverse effect in connection with the recall, but those who are prescribed with Accord’s hydrochlorothiazide should make sure the pills are a light orange color with a letter H on one side, and a number 1 on the other. Consumers are asked to report any drugs that do not match the description and check with their pharmacist. If you have any questions you can also contact Accord Healthcare at 1-855-869-1081 or [email protected].