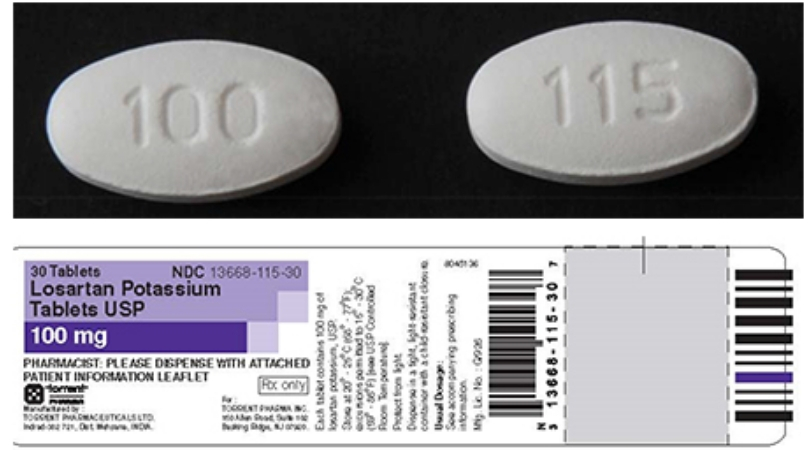
A pharmaceutical company has issued a voluntary recall of blood pressure medication. According to the FDA, Torrent Pharmaceuticals limited is voluntarily recalling two lots of Losartan potassium tablets.
Videos by Rare
Medical officials say the medication is being recalled after experts noticed detection of trace amounts of an “unexpected impurity” found in an active pharmaceutical ingredient (API) manufactured by Hetero Labs Limited.
The impurity detected in that API is N-nitrosodimethylamine, also known as NDEA, a substance that occurs naturally in certain foods, air pollution, drinking water, and industrial processes.
The impurity has been classified as a probable human carcinogen as per International Agency for Research on Cancer (IARC) classification. To date, Torrent Pharmaceuticals Limited has stated they have not received any reports of reactions or incidents related to the recall.
Losartan is usually used to treat high blood pressure, hypertension, hypertensive patients with Left Ventricular Hypertrophy and for treatment of nephropathy in Type 2 diabetic patients.
The following products recalled are packaged in bottles, listed below. The product can be identified by checking the product name, manufacturer details, and bath or lot number the bottle containing these products.
- LOSARTAN POTASSIUM TAB, USP 100mg, 30-count bottles, lot/batch BO31C016, expatriation date 04/2019.
- LOSARTAN POTASSIUM TAB, USP 100mg, 90-count bottles, lot/batch BO31C016, expiration date 04/2019.
- LOSARTAN POTASSIUM TAB, USP 100mg,1000-count bottles, lot/batch 4DK3C005, expiration date 04/2019.
If you have any question about the products or about the recall you are asked to contact Torrent Pharmaceuticals Limited at 1-800-912-9561 or visit the Food and Drug Administration’s website.